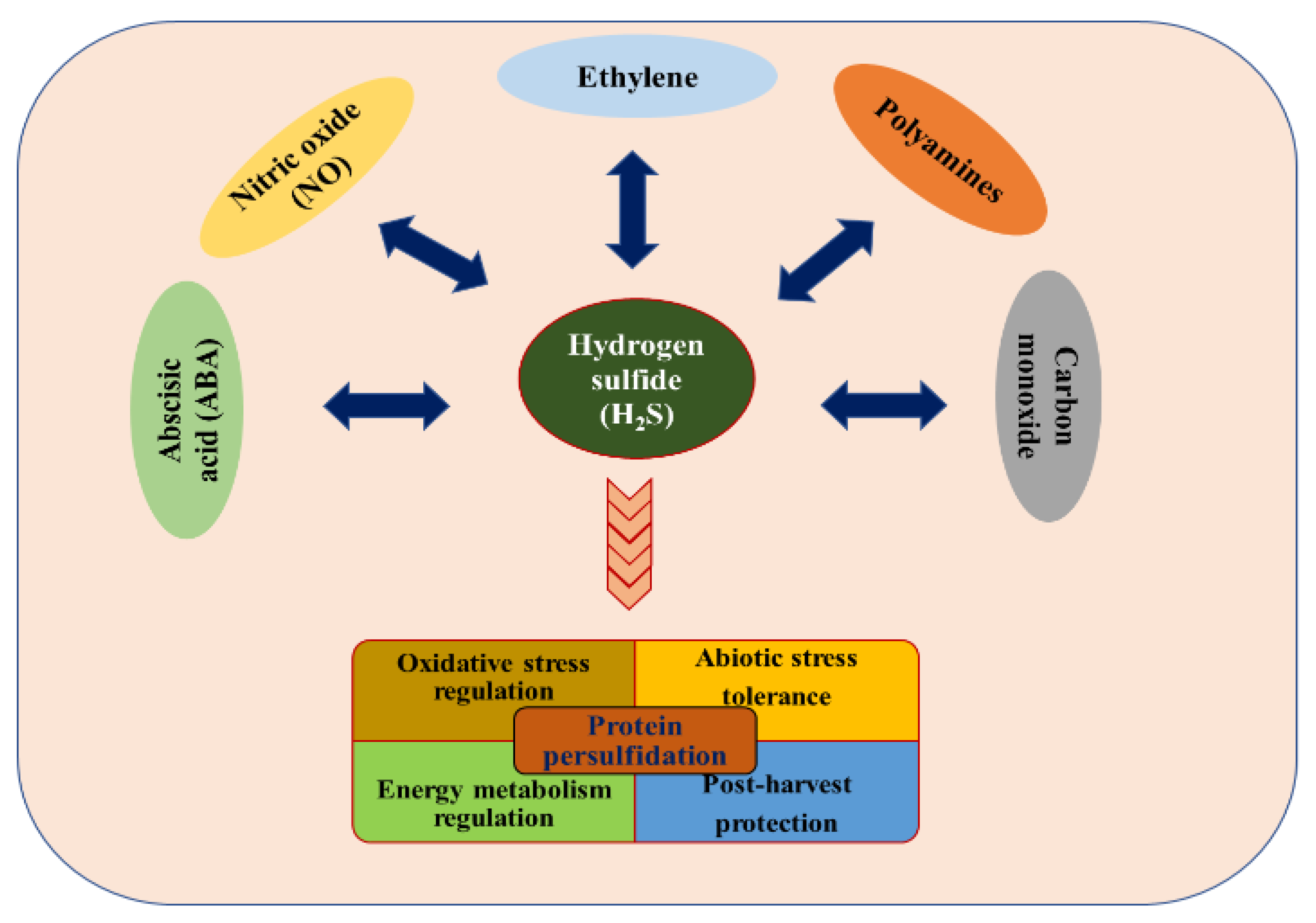
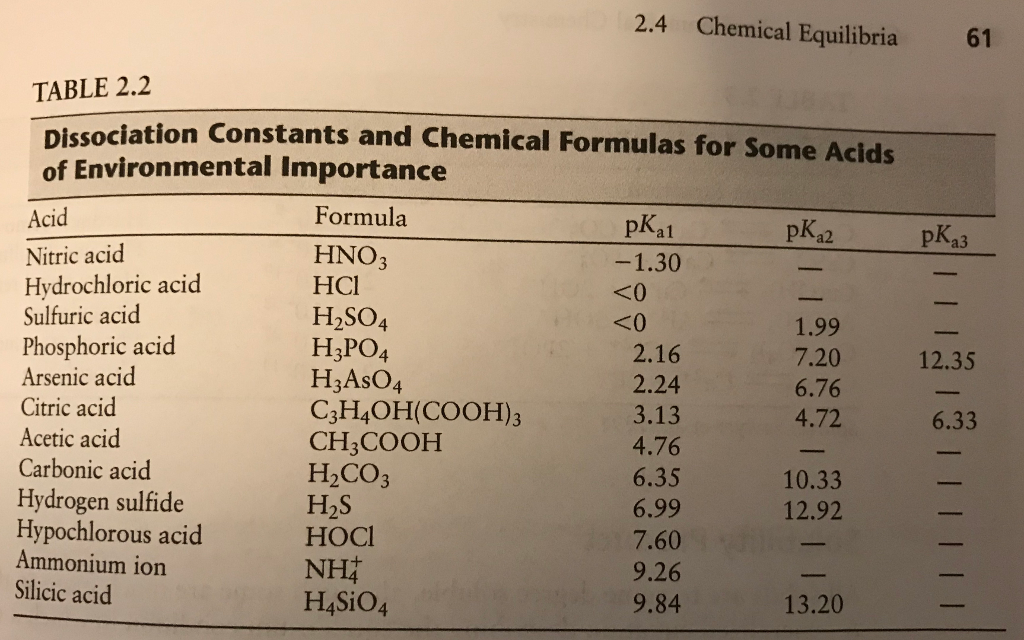
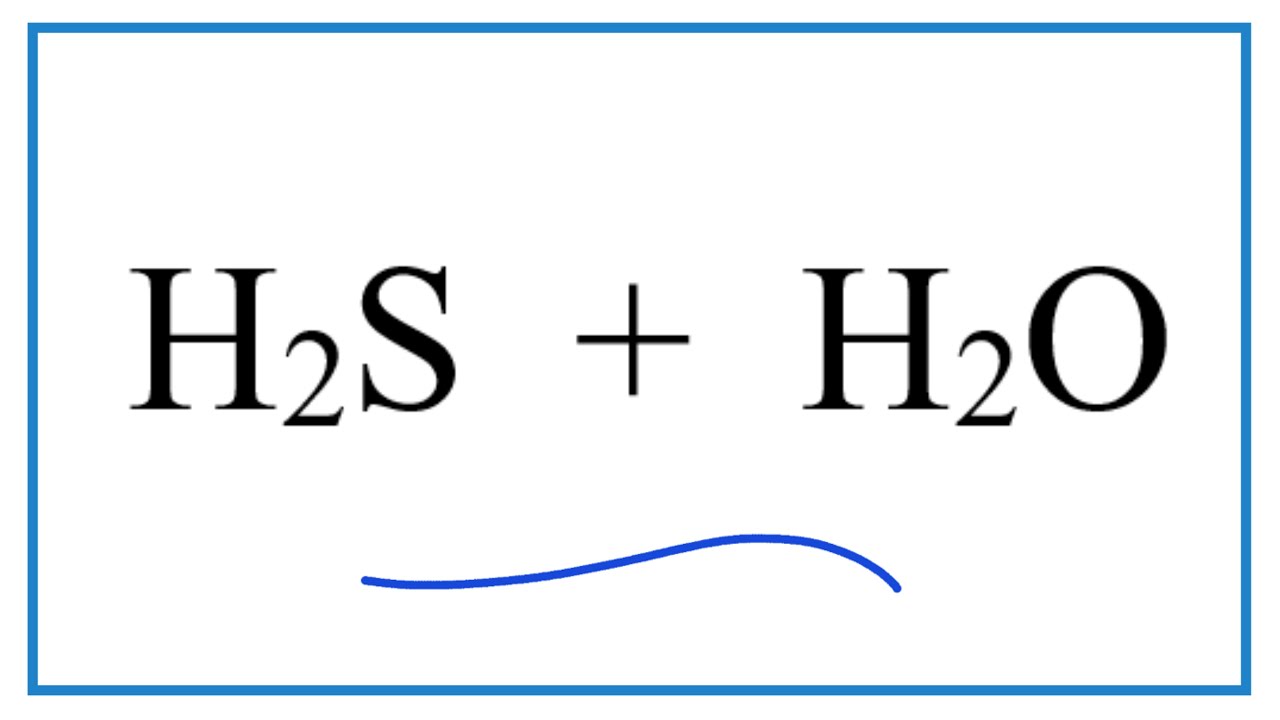Experts please tell whether H2S is lewis acid or lewis base and bronsted acid or bronsted base - Chemistry - Hydrogen - 16684677 | Meritnation.com

Will the following reaction favor the forward reaction or favor the reverse reaction? Explain. HF(aq) + HS-(aq) arrow F-(aq) + H2S(aq) | Homework.Study.com

Acid-base reaction with H2S liberation versus time. H2S detection via... | Download Scientific Diagram
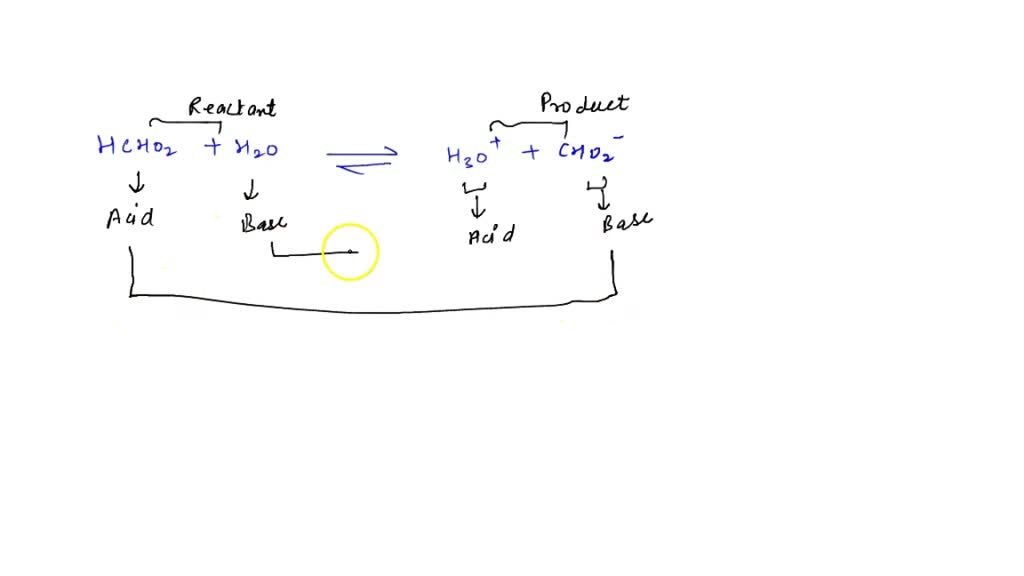
SOLVED: In aqueous solution, HS- is the conjugate acid of and the conjugate base of . Group of answer choices H2S, S2- H3O+, S2- S2-, H2S S2-, H3O+
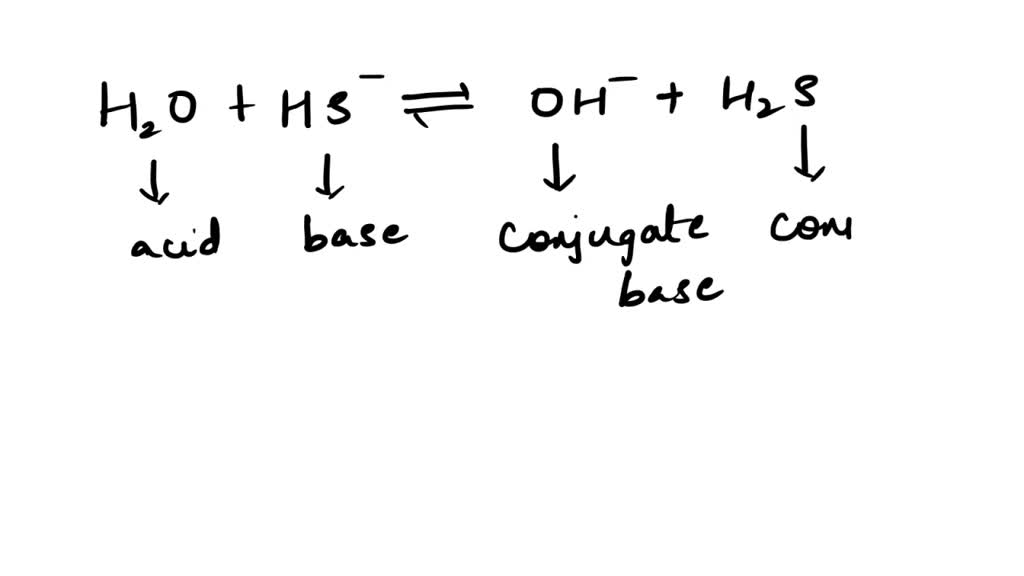
SOLVED: Be sure to answer all parts. In the following equation, identify the acids, bases, and conjugate pairs. H2O + HS− ⇌ OH− + H2S (a) What is the acid? H2O HS−

Which of the species from the equilibrium below are conjugate acid-base pairs? NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com