Problem Set #10 Assigned November 8, 2013 – Due Friday, November 15, 2013 Please show all work for credit To Hand in 1.
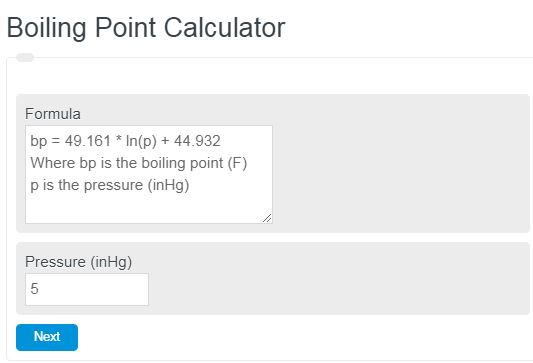
How does one calculate the boiling point of water at different pressures without a steam table? - Quora

Methanethiol has a vapor pressure of 429 torr at −25 ∘c and a normal boiling point of 6.0 ∘c. find δhvap - Brainly.com

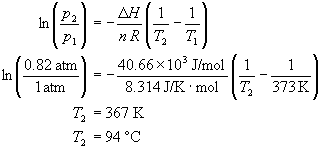
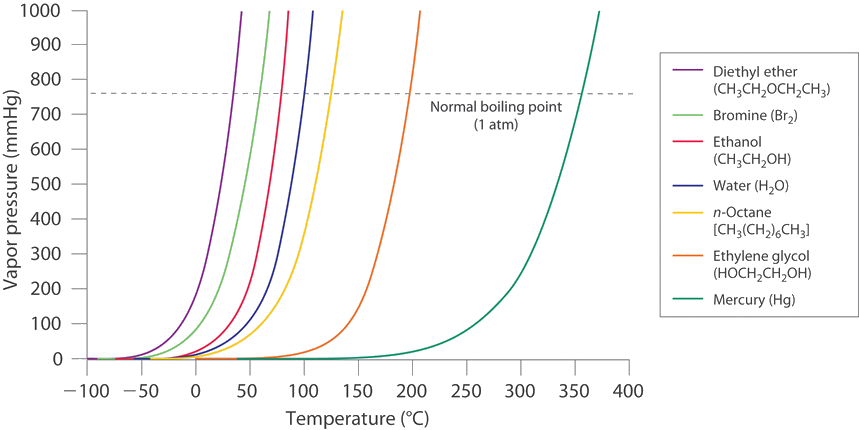
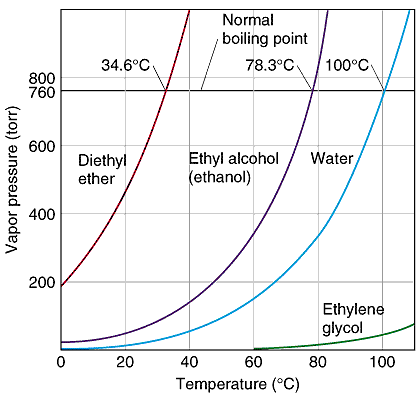
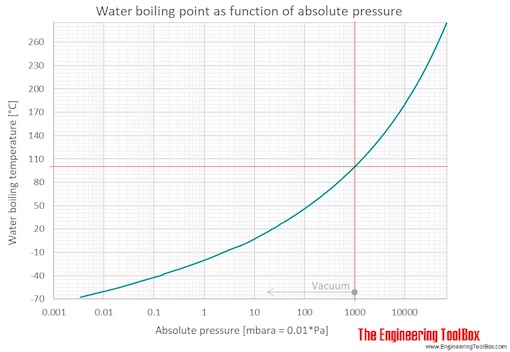




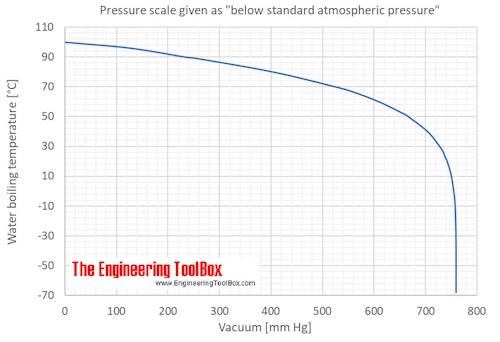
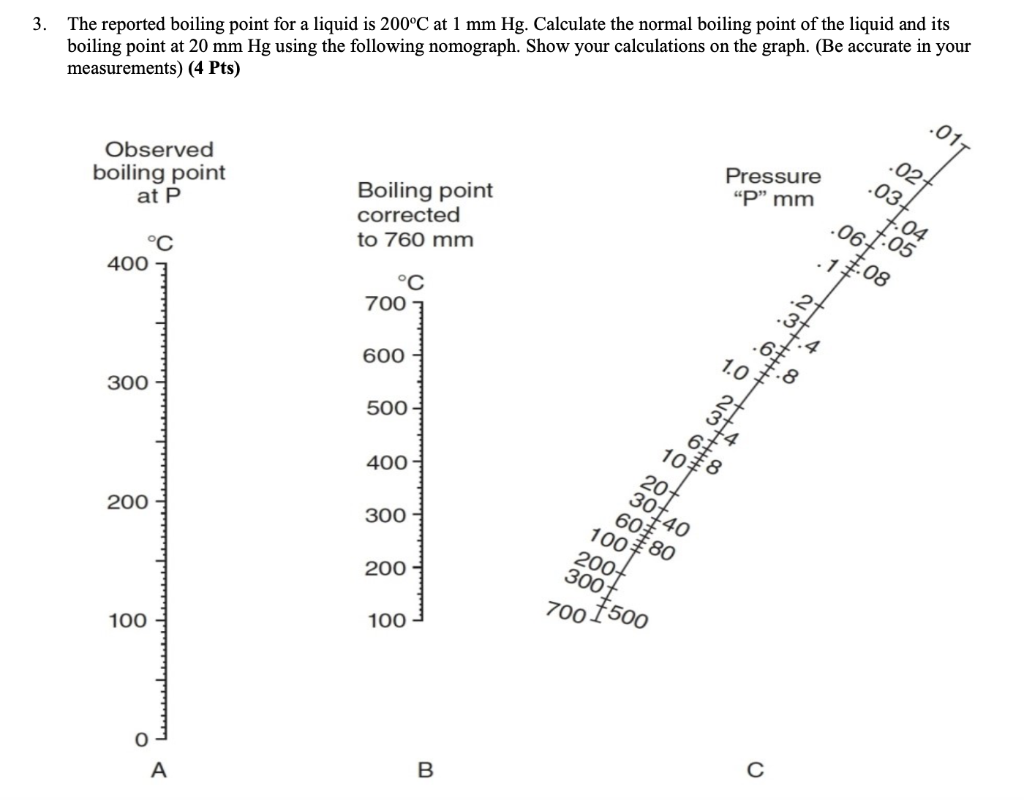

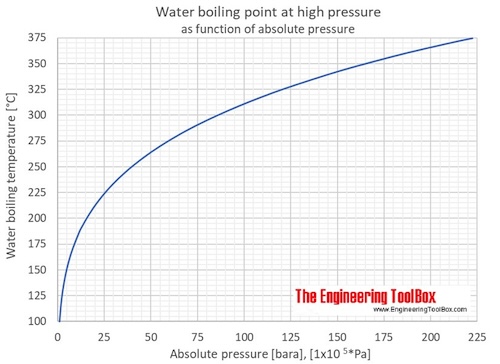

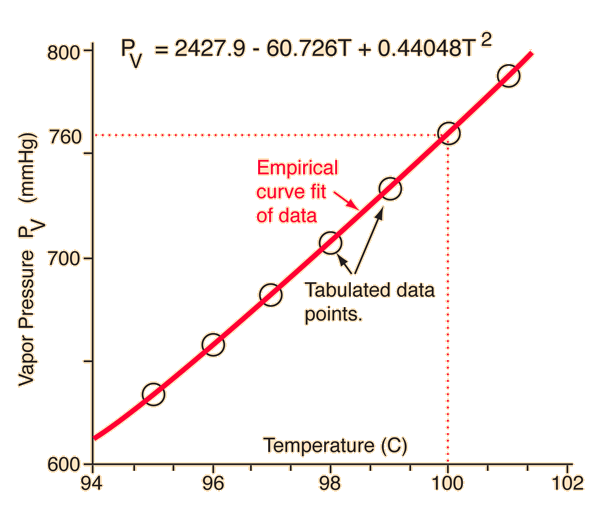

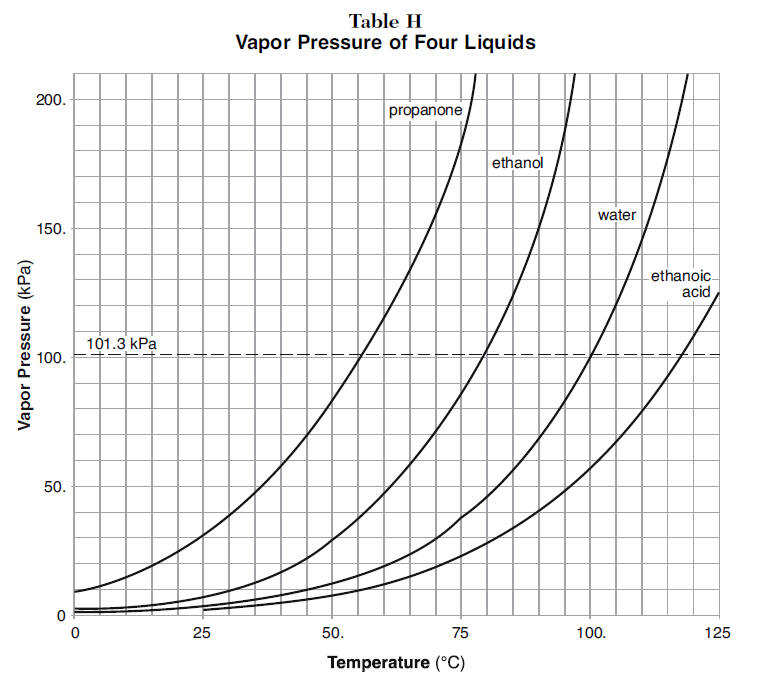


![Nomograph for the calculation of boiling points under vacuum - [www.rhodium.ws] Nomograph for the calculation of boiling points under vacuum - [www.rhodium.ws]](https://www.designer-drug.com/pte/12.162.180.114/dcd/chemistry/equipment/pictures/nomograph.gif)