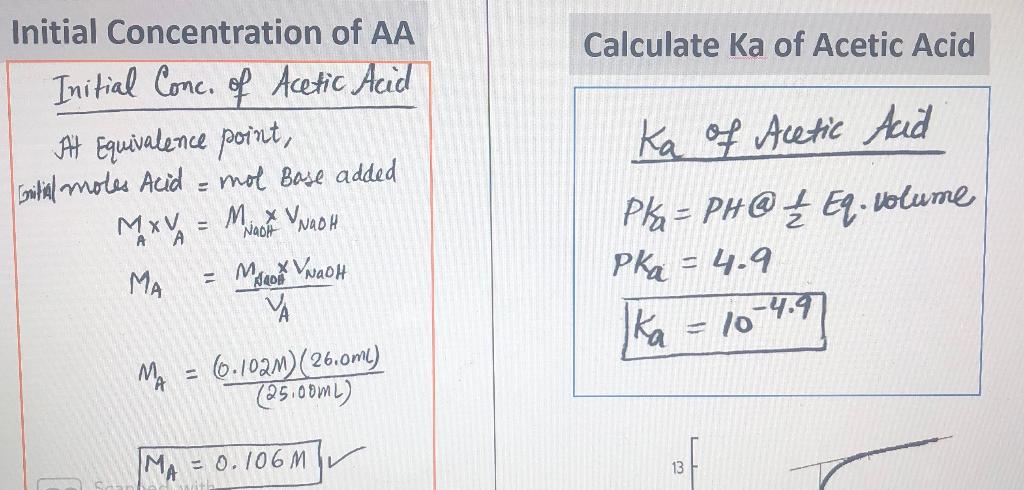
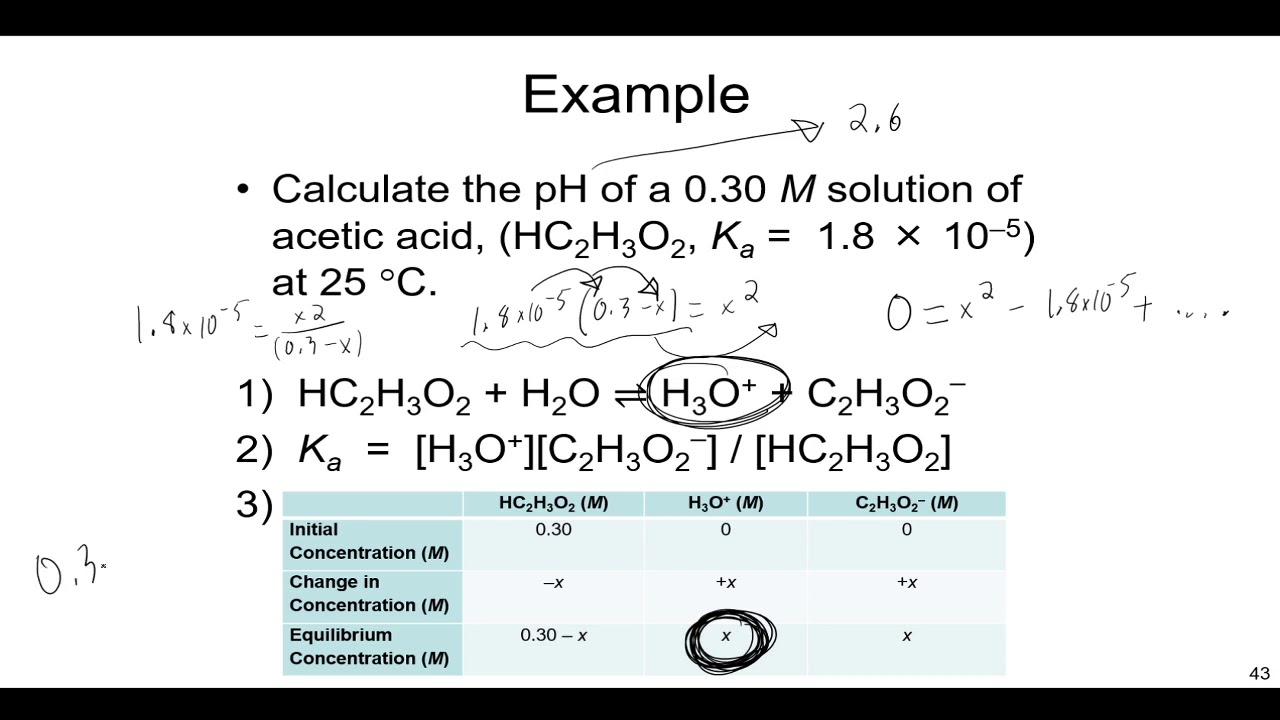
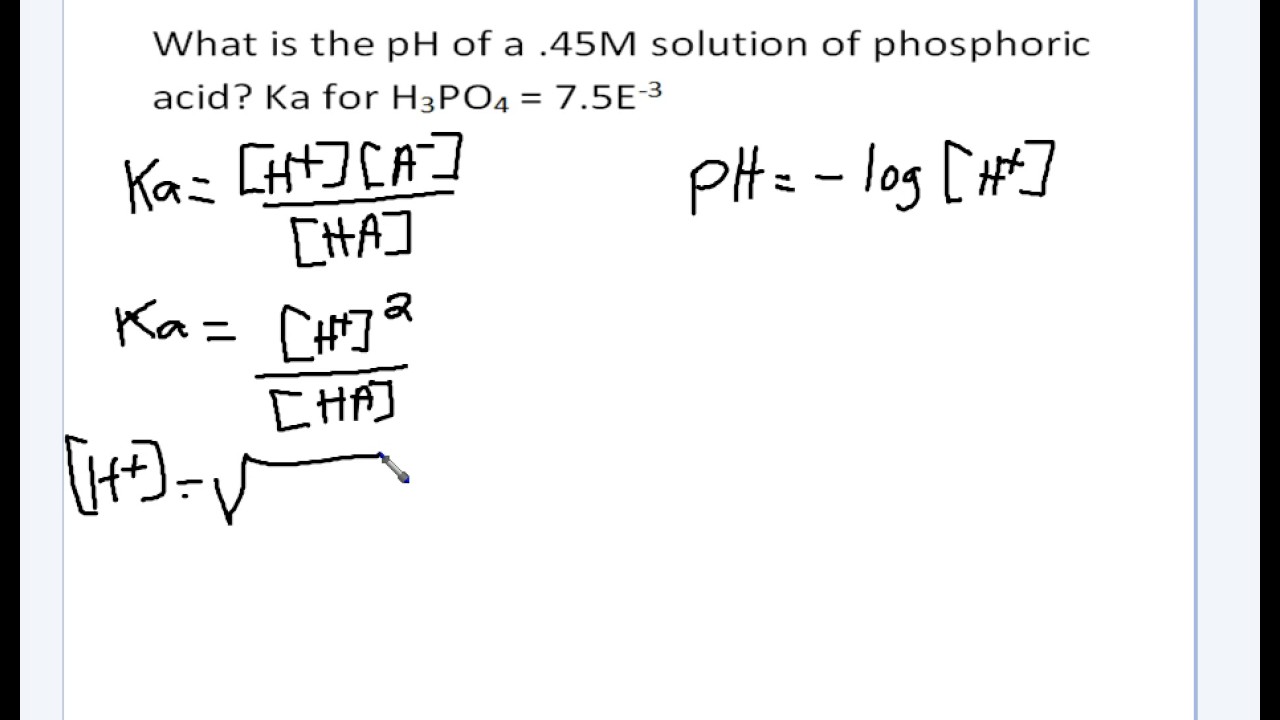
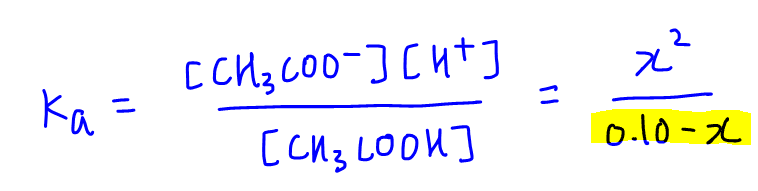The Ka value for acetic acid, CH3COOH(aq), is 1.8x10^-5. Calculate the ph of a 2.80 M acetic acid solution - Home Work Help - Learn CBSE Forum
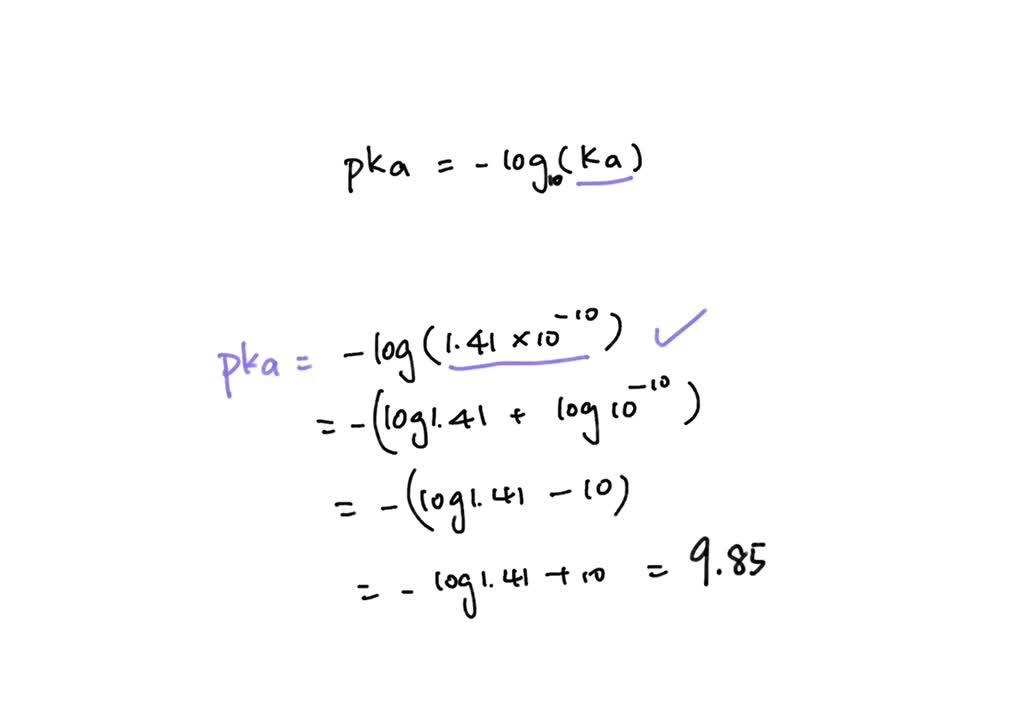
SOLVED: How would you calculate the pka or ka for CH2ClCH2COOH and CH2FCH2COOH if you only knew that this is a carboxylic acid? Do you write the conjugate acid and base to

A 0.21 M solution of chloroacetic acid, ClCH2CO2H, has a pH of 1.79. Calculate Ka for the acid. | Homework.Study.com